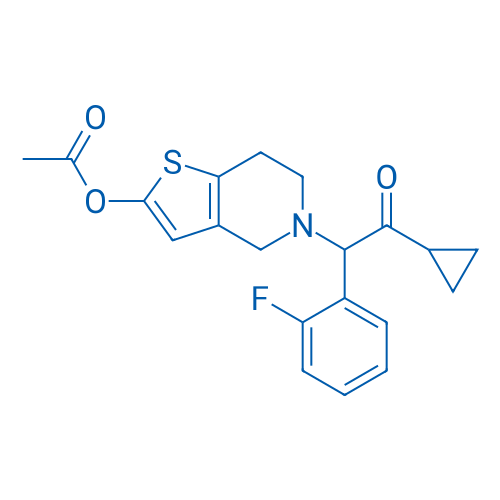
CAS No.: 150322-43-3
普拉格雷 Catalog No. CSN16171
Synonyms: PCR 4099;CS-747;LY-640315
Prasugrel is a platelet inhibitor with IC50 value of 1.8 μM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01276275 - - Completed - Sweden ... more >> Research Site Stockholm, Sweden Collapse << NCT03430661 Healthy Phase 1 Recruiting June 30, 2019 United States, New Jersey ... more >> Biotrial Inc Recruiting Newark, New Jersey, United States, 07103 Collapse << NCT00830960 Acute Coronary Syndrome Phase 3 Completed - China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100853 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guang Zhou, China, 510080 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310009 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanjing, China, 210008 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China, 200433 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shenyang, China, 110016 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wenzhou, China, 325027 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xi'An, China, 710061 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Daegu, Korea, Republic of, 700-721 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kwang Ju, Korea, Republic of, 501-757 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seongnam-Si, Korea, Republic of, 463-707 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 135 720 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Suwon-City, Korea, Republic of, 442-721 Taiwan For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taichung City, Taiwan, 40201 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taichung, Taiwan, 404 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taipei, Taiwan, 112 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tao-Yuan, Taiwan, 333 Thailand For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bangkok, Thailand, 10400 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiang Mai, Thailand, 50200 Collapse << - 更多
- 参考文献
- [1] Schror K, Siller-Matula JM, et al. Pharmacokinetic basis of the antiplatelet action of prasugrel. Fundam Clin Pharmacol. 2012 Feb;26(1):39-46.
- [2] Darius H. [Prasugrel] . Hamostaseologie. 2012;32(3):186-90.
- [3] M Cattaneo, P2Y12 receptors: structure and function. J Thromb Haemost. 2015 Jun;13 Suppl 1:S10-6.
- [4] Wijeyeratne YD, et al. Anti-platelet therapy: ADP receptor antagonists. Br J Clin Pharmacol. 2011 Oct;72(4):647-57.
- [5] Sugidachi A, et al. The greater in vivo antiplatelet effects of prasugrel as compared to clopidogrel reflect more efficient generation of its active metabolite with similar antiplatelet activity to that of clopidogrel's active metabolite. J Thromb Haemost. 2007 Jul;5(7):1545-51.
- [6] Asmaa A Gomaa, Hanan S El-Abhar, et al. Prasugrel anti-ischemic effect in rats: Modulation of hippocampal SUMO2/3-IкBα/Ubc9 and SIRT-1/miR-22 trajectories. Toxicol Appl Pharmacol. 2021 Sep 1;426:115635.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 150322-43-3 | 储存条件 |
|
|||||||||||||
| 分子式 | C20H20FNO3S | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 373.44 | 别名 | PCR 4099;CS-747;LY-640315;Effient;普拉格雷 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01276275 | - | - | Completed | - | Sweden ... more >> Research Site Stockholm, Sweden Collapse << |
| NCT03430661 | Healthy | Phase 1 | Recruiting | June 30, 2019 | United States, New Jersey ... more >> Biotrial Inc Recruiting Newark, New Jersey, United States, 07103 Collapse << |
| NCT00830960 | Acute Coronary Syndrome | Phase 3 | Completed | - | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100853 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guang Zhou, China, 510080 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310009 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanjing, China, 210008 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China, 200433 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shenyang, China, 110016 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wenzhou, China, 325027 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xi'An, China, 710061 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Daegu, Korea, Republic of, 700-721 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kwang Ju, Korea, Republic of, 501-757 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seongnam-Si, Korea, Republic of, 463-707 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 135 720 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Suwon-City, Korea, Republic of, 442-721 Taiwan For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taichung City, Taiwan, 40201 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taichung, Taiwan, 404 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taipei, Taiwan, 112 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tao-Yuan, Taiwan, 333 Thailand For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bangkok, Thailand, 10400 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiang Mai, Thailand, 50200 Collapse << |
| NCT01765400 | Systolic Heart Failure | Phase 4 | Completed | - | United States, Nebraska ... more >> University of Nebaska Medical Center Omaha, Nebraska, United States, 68198 Collapse << |
| NCT01107899 | Acute Coronary Syndromes | Phase 1 | Terminated(Terminated due to E... more >>nrollment futility) Collapse << | - | Germany ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Munich, Germany, 80636 Collapse << |
| NCT02032303 | Acute Coronary Syndrome ... more >> Non ST Elevation Myocardial Infarction ST Elevation Myocardial Infarction Unstable Angina Collapse << | Phase 4 | Unknown | - | Italy ... more >> Massimo Mancone Not yet recruiting Rome, Italy, 00100 Contact 00390649970468 Principal Investigator: massimo mancone Collapse << |
| NCT00357968 | Coronary Artery Disease | Phase 2 | Completed | - | United States, Florida ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jacksonville, Florida, United States, 32209 United States, Massachusetts For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Worcester, Massachusetts, United States, 01655 United States, Michigan For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ann Arbor, Michigan, United States, 48109 United States, South Dakota For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rapid City, South Dakota, United States, 57701 France For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lille, France, 59037 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Marseille, France, 13385 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Paris, France, 75013 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tours, France, 37044 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bad Krozingen, Germany, D-79189 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Berlin, Germany, D-12203 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Giessen, Germany, 35392 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. München, Germany, D-80636 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tubingen, Germany, 72076 Israel For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Haifa, Israel, 31096 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jerusalem, Israel, 91120 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tel Hashomer, Israel, 52621 Collapse << |
| NCT01107899 | - | - | Terminated(Terminated due to E... more >>nrollment futility) Collapse << | - | - |
| NCT02422888 | Myocardial Infarction | Phase 4 | Active, not recruiting | October 2019 | Netherlands ... more >> VU University Medical Center Amsterdam, Noord-Holland, Netherlands, 1081 HV Collapse << |
| NCT00357968 | - | - | Completed | - | - |
| NCT00830960 | - | - | Completed | - | - |
| NCT01090336 | - | - | Unknown | December 2011 | Germany ... more >> University of Heidelberg Recruiting Heidelberg, Baden-Württemberg, Germany, 69120 Contact: Evangelos Giannitsis, Prof. Dr. +49 (0)6221-56-8611 Evangelos_Giannitsis@med.uni-heidelberg.de Sub-Investigator: Kerstin Kurz, Dr. Collapse << |
| NCT02317198 | Non-ST-elevation Acute Coronar... more >>y Syndrome Unstable Angina Non-ST-elevation Myocardial Infarction Collapse << | Phase 4 | Recruiting | October 2019 | Netherlands ... more >> St. Antonius hospital Recruiting Nieuwegein, Utrecht, Netherlands, 3430 EM Collapse << |
| NCT02487732 | Acute Coronary Syndrome | Phase 4 | Unknown | - | Korea, Republic of ... more >> Korea University Anam Hospital Seoul, Korea, Republic of, 136-705 Collapse << |
| NCT00059215 | - | - | Completed | - | - |
| NCT00356135 | Coronary Arteriosclerosis ... more >> Acute Coronary Syndrome Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Monday-Friday from 9:00 AM to 5:00 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Jacksonville, Florida, United States, 32209 United States, Maryland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Monday-Friday from 9:00 AM to 5:00 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Baltimore, Maryland, United States, 21215 United States, Massachusetts For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Monday-Friday from 9:00 AM to 5:00 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Worcester, Massachusetts, United States, 01655 United States, Michigan For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Monday-Friday from 9:00 AM to 5:00 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Ann Arbor, Michigan, United States, 48109 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Monday-Friday from 9:00 AM to 5:00 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Cincinnati, Ohio, United States, 45219 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Monday-Friday from 9:00 AM to 5:00 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Columbus, Ohio, United States, 43210 United States, Oklahoma For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Monday-Friday from 9:00 AM to 5:00 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Oklahoma City, Oklahoma, United States, 73104 United States, South Dakota For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Monday-Friday from 9:00 AM to 5:00 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Rapid City, South Dakota, United States, 57701 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Monday-Friday from 9:00 AM to 5:00 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Houston, Texas, United States, 77024 Collapse << |
| NCT00642174 | Diabetes Mellitus ... more >> Coronary Artery Disease Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jacksonville, Florida, United States, 32209 United States, Massachusetts For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Worcester, Massachusetts, United States, 01655 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. New York, New York, United States, 10029 United States, Oklahoma For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Oklahoma City, Oklahoma, United States, 73104 Collapse << |
| NCT01365741 | Heart Disease ... more >> Ischemic Heart Disease ST-elevation Myocardial Infarction Collapse << | Not Applicable | Completed | - | Denmark ... more >> Nordsjællands Hospital Hillerød, Denmark, 2100 Collapse << |
| NCT00642174 | - | - | Completed | - | - |
| NCT00059215 | Cardiovascular Diseases ... more >> Heart Diseases Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST) or speak with your personal physician. Boston, Massachusetts, United States Canada, British Columbia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4599) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Victoria, British Columbia, Canada Collapse << |
| NCT00356135 | - | - | Completed | - | - |
| NCT00097591 | Coronary Arteriosclerosis ... more >> Acute Coronary Syndromes Collapse << | Phase 3 | Completed | - | United States, Indiana ... more >> For more information regarding investigative sites for this trial, call 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri 9 AM - 5 PM Eastern Time (UTC/GMT - 5 hours, EST), Global Quintiles Study Line (1-866-615-4672) or speak with your physician Indianapolis, Indiana, United States Collapse << |
| NCT02548611 | Angina Pectoris | Phase 4 | Recruiting | November 2019 | Germany ... more >> Universitäts-Herzzentrum Freiburg, Bad Krozingen Recruiting Freiburg, Bad Krozingen, Germany, 79189 Contact: Willibald Hochholzer, MD +497633402 ext 5212 Willibald.Hochholzer@universitaets-herzzentrum.de Contact: Ulrike Maier +497633402 ext 5212 Ulrike.Maier@universitaets-herzzentrum.de Munich University Hospital Recruiting Munich, Bavaria, Germany, 81377 Contact: Julinda Mehilli, MD Contact: Janina Neubarth-Mayer +49 89 44007 ext 3141 Janina.Neubarth@med.uni-muenchen.de Deutsches Herzzentrum Muenchen Recruiting Munich, Germany, 80636 Contact: Adnan Kastrati, MD +49 89 1218 ext 4578 kastrati@dhm.mhn.de Contact: Jens Wiebe, MD +49 89 1218 ext 1514 wiebe@dhm.mhn.de Principal Investigator: Adnan Kastrati, MD Sub-Investigator: Jens Wiebe, MD Klinikum Bogenhausen Recruiting Munich, Germany Contact: Markus Deichstetter, MD 49 89 9270 ext 702770 Markus.Deichstetter@klinikum-muenchen.de Contact: Christian Tesche, MD 49 89 9270 ext 702770 Christian.Tesche@klinikum-muenchen.de Principal Investigator: Markus Deichstetter, MD Sub-Investigator: Christian Tesche, MD Hungary Heart Center Balatonfüred and Heart and Vascular Center Recruiting Balatonfüred, Hungary Contact: Daniel Aradi, MD +36 302355639 daniel_aradi@yahoo.com Principal Investigator: Daniel Aradi, MD Collapse << |
| NCT00699998 | Acute Coronary Syndrome | Phase 3 | Completed | - | - |
| NCT00097591 | - | - | Completed | - | - |
| NCT00995514 | - | - | Terminated(Administrative reas... more >>ons) Collapse << | - | United States, New Jersey ... more >> Medco Health Solutions, Inc Franklin Lakes, New Jersey, United States, 07417 Collapse << |
| NCT01135667 | Coronary Artery Disease ... more >> Percutaneous Coronary Intervention Acute Coronary Syndromes Collapse << | Phase 4 | Completed | - | Denmark ... more >> Rigshospitalet, Department of Cardiology, 2013 Copenhagen O, Denmark, 2100 Collapse << |
| NCT01346800 | Healthy Volunteers | Phase 1 | Completed | - | Switzerland ... more >> University Hospitals Geneva 14, Switzerland, 1211 Collapse << |
| NCT01015287 | Acute Coronary Syndromes | Phase 3 | Completed | - | - |
| NCT01777503 | Acute Coronary Syndrome ... more >> Coronary Arteriosclerosis Myocardial Ischemia Cardiovascular Diseases Collapse << | Phase 4 | Unknown | December 2017 | Italy ... more >> Azienda Ospedaliera "Ospedale Civile di Legnano" Recruiting Legnano, MI, Italy, 20025 Contact: Stefano De Servi, MD stefano.deservi@ao-legnano.it Principal Investigator: Stefano De Servi, MD Arcispedale Santa Maria Nuova- IRCCS Recruiting Reggio Emilia, Italy, 42123 Contact: Stefano Savonitto, MD +39-0341-489490 s.savonitto@ospedale.lecco.it Principal Investigator: Stefano Savonitto, MD Collapse << |
| NCT00976092 | Myocardial Infarction | Phase 4 | Unknown | February 2015 | Germany ... more >> Deutsches Herzzentrum Muenchen Munich, Bavaria, Germany, 80636 Klinikum rechts der Isar, Technische Universitaet Muenchen Munich, Bavaria, Germany, 81674 Herzzentrum der Segeberger Kliniken Bad Segeberg, Germany, 23795 Collapse << |
| NCT01336348 | ST Segment Elevation Myocardia... more >>l Infarction Collapse << | Phase 3 | Completed | - | Italy ... more >> Cardiology Unit Ferrara, Italy, 44100 Collapse << |
| NCT00699998 | - | - | Completed | - | - |
| NCT01015287 | - | - | Completed | - | - |
| NCT01107912 | Coronary Artery Disease | Phase 1 | Completed | - | United States, Florida ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jacksonville, Florida, United States, 32209 United States, Maryland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Baltimore, Maryland, United States, 21215 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cincinnati, Ohio, United States, 45212 Ireland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dublin, Ireland, 9 Netherlands For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nieuwegein, Netherlands, 3435 CM Sweden For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lund, Sweden, 22185 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Malmo, Sweden, 20502 Collapse << |
| NCT01360437 | Coronary Artery Disease ... more >> Acute Coronary Syndrome Collapse << | Phase 3 | Completed | - | Greece ... more >> Cardiology Department Patras University Hospital Rio, Patras, Greece, 26500 Collapse << |
| NCT01107925 | - | - | Completed | - | - |
| NCT00910299 | - | - | Terminated(Due to the low rate... more >> of primary endpoint events experienced in the study to date) Collapse << | - | - |
| NCT01107912 | - | - | Completed | - | - |
| NCT01107925 | Coronary Artery Disease | Phase 1 | Completed | - | United States, Florida ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jacksonville, Florida, United States, 32209 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cincinnati, Ohio, United States, 45212 Ireland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dublin, Ireland, 9 Netherlands For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nieuwegein, Netherlands, 3435 CM Sweden For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lund, Sweden, 22185 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Uppsala, Sweden, 75185 Collapse << |
| NCT01597375 | Asthma, Aspirin-Induced ... more >> Aspirin Exacerbated Asthma Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Asthma Research Center Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00910299 | Coronary Artery Disease (CAD) | Phase 2 | Terminated(Due to the low rate... more >> of primary endpoint events experienced in the study to date) Collapse << | - | United States, Florida ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Clearwater, Florida, United States, 33756 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jacksonville, Florida, United States, 32209 United States, Georgia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rome, Georgia, United States, 30165 United States, Illinois For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moline, Illinois, United States, 61265 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. New York, New York, United States, 10021 United States, Oregon For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Portland, Oregon, United States, 97225 United States, Pennsylvania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Pittsburgh, Pennsylvania, United States, 15213 United States, South Dakota For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rapid City, South Dakota, United States, 55701 United States, Tennessee For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nashville, Tennessee, United States, 37203 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bad Berka, Germany, 99437 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bad Krozingen, Germany, 79189 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bad Segeberg, Germany, 23795 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Berlin, Germany, 12203 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bremen, Germany, 28277 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dortmund, Germany, 44137 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Essen, Germany, 45147 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Freiburg, Germany, 79106 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fulda, Germany, 36043 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hamburg, Germany, 20246 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Leipzig, Germany, 04289 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mainz, Germany, 55131 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Munich, Germany, 80636 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Pforzheim, Germany, 75175 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Stuttgart, Germany, 70376 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tuebingen, Germany, 72076 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Villingen-Schwenningen, Germany, 78050 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wuppertal, Germany, 42117 Collapse << |
| NCT01597375 | - | - | Completed | - | - |
| NCT01642940 | Coronary Artery Disease ... more >> Acute Coronary Syndrome Collapse << | Phase 4 | Completed | - | Greece ... more >> Cardiology Department Patras University Hospital Patras, Achaia, Greece, 26500 Collapse << |
| NCT02944123 | Acute Coronary Syndrome | Phase 3 | Recruiting | February 2019 | Korea, Republic of ... more >> Dong-A University Hospital Recruiting Busan, Korea, Republic of, 602-715 Contact: Moo Hyun Kim, MD +82-51-240-2976 kimmh@dau.ac.kr Collapse << |
| NCT01103843 | Coronary Artery Disease ... more >> Platelet Aggregation Inhibitors PCI- Percutaneous Coronary Intervention Collapse << | Not Applicable | Unknown | May 2011 | United States, New York ... more >> St. Francis Hospital Recruiting Roslyn, New York, United States, 11576 Contact: Elizabeth S Haag, RN, MPA,CCRP 516-562-6790 elizabeth.haag@chsli.org Contact: Lyn Santiago, RN,CCRC 516 562-6790 lyn.santiago@chsli.org Principal Investigator: Richard A Shlofmitz, MD Collapse << |
| NCT03435133 | ST Elevation Myocardial Infarc... more >>tion Collapse << | Not Applicable | Recruiting | December 31, 2018 | Spain ... more >> Pedro Dorado Recruiting Salamanca, Spain, 37007 Contact: Pedro Dorado 3492329100 ext 55369 pedro.dorado@gmail.com Collapse << |
| NCT01642966 | Acute Coronary Syndrome | Phase 4 | Completed | - | Greece ... more >> Cardiology Department Patras University Hospital Patras, Achaia, Greece, 26500 Collapse << |
| NCT03016611 | Acute Coronary Syndrome ... more >> STEMI Collapse << | Phase 4 | Not yet recruiting | - | Israel ... more >> Sheba Medical center Ramat Gan, Israel Collapse << |
| NCT00385944 | Acute Coronary Syndrome | Phase 2 | Completed | - | France ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Creteil, France, 94010 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Paris, France, 75013 Collapse << |
| NCT01158846 | ST-Elevation Myocardial Infarc... more >>tion Primary Percutaneous Coronary Intervention Collapse << | Phase 4 | Unknown | June 2011 | Italy ... more >> Istituto Clinico S. Ambrogio Recruiting Milan, Italy, 20149 Contact: Luca Testa, MD, PhD +39-3490808660 luctes@gmail.com Principal Investigator: Luca Testa, Md, PhD Principal Investigator: Francesco Bedogni, MD Collapse << |
| NCT00385944 | - | - | Completed | - | - |
| NCT01538446 | Acute Coronary Syndrome | Phase 4 | Completed | - | France ... more >> CHU Caremeau à Nimes - Service de Cardiologie Nimes, France, 30029 ACTION study group - Institut de Cardiologie- Hôpital la Pitié Salpêtrière Paris, France, 75013 Collapse << |
| NCT01338909 | Myocardial Infarction | Phase 3 | Completed | - | Greece ... more >> Patras University Hospital Patras, Greece, 26500 Collapse << |
| NCT01959451 | Acute Coronary Syndrome | Phase 4 | Completed | - | Austria ... more >> Medizin-Universität Graz, Univ. Klinik für Innere Medizin Graz, Austria, 8036 Wilhelminenspital Wien, 3. Medizinische Abteilung Vienna, Austria, 1160 Germany Klinikum Augsburg, Department of Cardiology Augsburg, Germany, 86156 Heart Center Bad Krozingen Bad Krozingen, Germany, 79189 Asklepios Stadtklinik Bad Tölz, Internal Medicine Bad Tölz, Germany, 83646 St. Josef Hospital, Katholisches Klinikum Bochum, Department of Cardiology Bochum, Germany, 44791 Universitätsklinikum Frankfurt, Department of Cardiology Frankfurt, Germany, 60590 Kliniken Ostallgäu-Kaufbeuren, Klinik Füssen Füssen, Germany, 87629 Heart Center at the University Medical Center Goettingen Goettingen, Germany, 37075 Universitätsmedizin Greifswald, Klinik u. Poliklinik für Innere Medizin B Greifswald, Germany, 17475 Universitäres Herzzentrum Hamburg, UKE Hamburg, Germany, 20246 Herzzentrum der Universität zu Köln Köln, Germany, 50937 University Hospital Mainz, Department of Cardiology Mainz, Germany, 55131 Klinikum Memmingen, Innere Medizin I Memmingen, Germany, 87700 Munich University Hospital Munich, Germany, 81377 Klinikum Neuperlach, Department of Cardiology Munich, Germany, 81737 Klinikum Bogenhausen, Department of Cardiology Munich, Germany, 81925 Klinikum Oldenburg gGmbH, Herz-Kreislauf-Zentrum, Klinik für Kardiologie Oldenburg, Germany, 26133 Universitätsmedizin Rostock, Zentrum für Innere Medizin Rostock, Germany, 18057 Helios-Klinikum Siegburg, Abteilung für Kardiologie und Angiologie Siegburg, Germany, 53721 University Hospital of Tuebingen, Department of Cardiology Tuebingen, Germany, 72076 Kliniken Nordoberpfalz AG, Klinikum Weiden, Medizinische Klinik II Weiden, Germany, 92637 Hungary Semmelweis Egyetem Kardiovaszkuláris Centrum Budapest, Hungary, 1122 Budapest Military Hospital Budapest, Hungary, 1134 Heart Center Balatonfüred Budapest, Hungary, 8230 Department of Cardiology Petz Aladár Megyei Oktató Kórház Györ, Hungary, 9023 Heart Center Kecskemet Kecskemet, Hungary, 6000 PTE KK Szívgyógyászati Klinika Intervenciós Kardiológia Részleg Pecs, Hungary, 7624 Heart Center Szeged Szeged, Hungary, 6720 Poland 3rd Department of Cardiology, Upper Silesian Medical Centre, Medical University of Silesia, Katowice Katowice, Poland, 40635 1st Department of Cardiology, Poznan University of Medical Science Poznan, Poland, 61848 1st Department of Cardiology, Medical University of Warsaw Warsaw, Poland, 2097 Department of Interventional Cardiology and Angiology, Institute of Cardiology, Warsaw Warsaw, Poland, 2795 Collapse << |
| NCT01305369 | Chronic Asthma | Phase 4 | Completed | - | Italy ... more >> Medicina 3 Ospedale San Paolo Dipartimento di Medicina Chirurgia e Odontoiatria, Università di Milano Milan, Italy, 20142 Collapse << |
| NCT01957540 | Coronary Artery Disease ... more >> Endothelial Function Collapse << | Phase 4 | Completed | - | Greece ... more >> Patras University Hospital Patras, Achaia, Greece, 26500 Collapse << |
| NCT01167023 | Sickle Cell Anemia | Phase 2 | Completed | - | United States, Alabama ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Birmingham, Alabama, United States, 35205 United States, Arkansas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Little Rock, Arkansas, United States, 72211 United States, California For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Sacramento, California, United States, 95817 United States, Florida For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Daytona Beach, Florida, United States, 32117 United States, Georgia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Augusta, Georgia, United States, 30912 United States, Indiana For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Indianapolis, Indiana, United States, 46260 United States, Maryland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Baltimore, Maryland, United States, 21205 United States, Massachusetts For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Boston, Massachusetts, United States, 02118 United States, Mississippi For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jackson, Mississippi, United States, 39216 United States, North Carolina For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chapel Hill, North Carolina, United States, 27599 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Greenville, North Carolina, United States, 27834 United States, Pennsylvania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jenkintown, Pennsylvania, United States, 19046 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Pittsburgh, Pennsylvania, United States, 15224 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Houston, Texas, United States, 77002 Collapse << |
| NCT01463163 | Platelet Reactivity | Phase 4 | Completed | - | Greece ... more >> Cardiology Department Patras University Hospital Rio, Achaia, Greece, 26500 Collapse << |
| NCT01109784 | Coronary Artery Disease (CAD) ... more >> Acute Coronary Syndrome (ACS) Collapse << | Phase 3 | Completed | - | Greece ... more >> Cardiology Department, Patras University Hospital Rio, Patras, Greece, 26500 Collapse << |
| NCT01463150 | Platelet Reactivity | Phase 4 | Completed | - | Greece ... more >> Cardiology Department Patras University Hospital Rio, Achaia, Greece, 26500 Collapse << |
| NCT01155765 | Hemodialysis ... more >>Chronic Renal Failure Collapse << | Phase 3 | Completed | - | Greece ... more >> Patras University Hospital Patras, Achaia, Greece, 26500 Collapse << |
| NCT01167023 | - | - | Completed | - | - |
| NCT01456364 | Coronary Heart Disease | Phase 4 | Unknown | May 2014 | Germany ... more >> Deutsches Herzzentrum Recruiting München, Bavaria, Germany, 80636 Contact: Katharina Mayer, MD +49-89-1218-2020 mayer.katharina@dhm.mhn.de Contact: Isabell Bernlochner, MD +49-89-4140-0 isabell.bernlochner@gmx.de Principal Investigator: Katharina Mayer, MD Sub-Investigator: Isabell Bernlochner, MD Klinikum der Ludwig-Maximilians-Universität München Recruiting München, Bavaria, Germany, 81377 Contact: Martin Orban, MD +49 89 7095 2371 martin.Orban@med.uni-muenchen.de Contact: Dirk Sibbing, MD +49 89 5160 2215 Dirk.Sibbing@med.uni-muenchen.de Principal Investigator: Martin Orban, MD Sub-Investigator: Dirk Sibbing, MD Hungary Heart Center Balatonfüred, Dept. of Cardiology Not yet recruiting Balatonfüred, Hungary, 8230 Contact: Daniel Aradi, MD +36302355639 daniel_aradi@yahoo.com Principal Investigator: Daniel Aradi, MD Sub-Investigator: Jozsef Faluközy, MD Collapse << |
| NCT02978040 | Coronary Artery Disease ... more >> STEMI - ST Elevation Myocardial Infarction Collapse << | Phase 4 | Recruiting | April 2019 | France ... more >> Group Hospitalier Pitié-Salpetrière Not yet recruiting Paris, France, 75013 Contact: Jean-Philippe Collet, Prof jean-philippe.collet@aphp.fr Italy University of Ferrara Not yet recruiting Ferrara, Italy, 44124 Contact: Gianluca Campo, Prof cmpglc@unife.it University of Naples Federico II Recruiting Naples, Italy, 80131 Contact: Giovanni Esposito, Prof espogiov@unina.it University of Sassari Not yet recruiting Sassari, Italy, 07100 Contact: Guido Parodi, Prof parodiguido@gmail.com Principal Investigator: Pierfranco Terrosu, MD Switzerland Bern University Hospital Recruiting Bern, Switzerland, 3010 Contact: Marco Valgimigli, Prof +41 316329653 marco.valgimigli@insel.ch Contact: Giuseppe Gargiulo, MD +41 316327205 giuseppe.gargiulo@insel.ch Collapse << |
| NCT01794000 | - | - | Terminated(The study is being ... more >>terminated for lack of efficacy.) Collapse << | - | - |
| NCT01304472 | Coronary Artery Disease (CAD) | Phase 3 | Completed | - | Greece ... more >> Dimitrios Alexopoulos Patras, Greece, 26500 Patras University Hospital Cardiology Department Patras, Greece, 26500 Collapse << |
| NCT01587651 | Coronary Artery Disease | Phase 4 | Completed | - | United States, Florida ... more >> Univ. of Florida College Medicine Jacksonville, Florida, United States, 32209 Clinical Pharmacology Unit of Miami Miami, Florida, United States, 33014 Progressive Medical Research Port Orange, Florida, United States, 32127 United States, Maryland Sinai Center for Thrombosis Research Baltimore, Maryland, United States, 21215 United States, Ohio Medpace Clinical Pharmacology Unit Cincinnati, Ohio, United States, 45212 United States, South Dakota Black Hills Cardiovascular Research Rapid City, South Dakota, United States, 57701 United States, Texas West Houston Area Clinical Trial Consultants Houston, Texas, United States, 77094 Cardiology Center of Houston Katy, Texas, United States, 77450 United Kingdom University Hospital of Wales Heath Park, Cardiff, United Kingdom, CF14 4XW Bristol Heart Institute Bristol, United Kingdom, B52 8HW University Hospital Leicester Leicester, United Kingdom, LE3 9QP Southampton General Hospital Southampton, United Kingdom, SO16 6YD New Cross Hospital Wolverhampton, United Kingdom, WV10 0QP Collapse << |
| NCT01944800 | Acute Coronary Syndrome (ACS) | Phase 4 | Recruiting | January 2021 | Germany ... more >> Universitäts-Herzzentrum Freiburg/ Bad Krozingen Recruiting Bad Krozingen, Baden-Württemberg, Germany, 79189 Contact: Franz-Josef Neumann, MD +49 7633 402-2000 franz-josef.neumann@universitaets-herzzentrum.de Contact: Barbara Steiger +49 7633 402-5204 Barbara.Steiger@universitaets-herzzentrum.de Principal Investigator: Franz-Josef Neumann, MD Sub-Investigator: Dietmar Trenk, MD Universitäts-Klinikum Heidelberg Recruiting Heidelberg, Baden-Württemberg, Germany, 69120 Contact: Hugo A. Katus, MD +49 6221 - 56 8670 hugo.katus@med.uni-heidelberg.de Contact: Heidi Deigentasch, SC Heidi.Deigentasch@med.uni-heidelberg.de Principal Investigator: Hugo A. Katus, MD Sub-Investigator: Evangelos Giannitsis, MD Universitätsklinikum Mannheim Recruiting Mannheim, Baden-Württemberg, Germany, 68167 Contact: Ibrahim Akin, MD +49 621 383-2204 Ibrahim.Akin@umm.de Contact: Stefanie Dorn, SC Stefanie.Dorn@umm.de Principal Investigator: Ibrahim Akin, MD Sub-Investigator: Michael Behnes, MD Universitätsklinikum Ulm Recruiting Ulm, Baden-Württemberg, Germany, 89081 Contact: Jochen Wöhrle, MD +49 731 500 450 28 (-01) jochen.woehrle@uniklinik-ulm.de Contact: Alexandra Maaß, SC +49 (0)731-500-45006 Alexandra.Maass@uniklinik-ulm.de Principal Investigator: Jochen Wöhrle, MD Sub-Investigator: Sinisa Markovic, MD Klinikum Landkreis Erding Recruiting Erding, Bavaria, Germany, 85435 Contact: Lorenz Bott-Flügel, MD lorenz.bott-fluegel@klinikum-erding.de Contact: Yvonne Anders, SN +49 8122 591613 yvonne.anders@klinikum-erding.de Principal Investigator: Lorenz Bott-Flügel, MD Sub-Investigator: Nader Joghetaei, MD Deutsches Herzzentrum Munich Recruiting München, Bavaria, Germany, 80636 Contact: Stefanie Schuepke, MD +49-89-1218 ext 0 schuepke@dhm.mhn.dee Contact: Deborah-Ann Schuster +49-89-1218 ext 1528 schuster@dhm.mhn.de Principal Investigator: Stefanie Schuepke, MD Sub-Investigator: Katharina Mayer, MD Klinikum rechts der Isar, 1. Medizinische Klinik und Poliklinik Recruiting München, Bavaria, Germany, 81675 Contact: Isabell Bernlochner, MD +49 89 4140 2350 isabell.bernlochner@mri.tum.de Principal Investigator: Karl-Ludwig Laugwitz, MD Sub-Investigator: Isabell Bernlochner, MD Klinikum Neuperlach Recruiting München, Bavaria, Germany, 81737 Contact: Harald Mudra, MD +49 89 6794-2351 harald.mudra@klinikum-muenchen.de Contact: Jacqueline Fiedler, SC +49 89 6794 2353 jacqueline.fiedler@klinikum-muenchen.de Principal Investigator: Harald Mudra, MD Sub-Investigator: Manuela Segerer, MD Universitätsklinikum Regensburg Recruiting Regensburg, Bavaria, Germany, 93042 Contact: Marcus Fischer, MD +49 941 944-7299 marcus.fischer@ukr.de Contact: Ingrid Lugauer, SC +49 941 944 7337 ingrid.lugauer@klinik.uni-regensburg.de Principal Investigator: Marcus Fischer, MD Sub-Investigator: Andrea Bässler, MD Klinikum Traunstein Active, not recruiting Traunstein, Bavaria, Germany, 83278 Kerckhoff-Klinik GmbH, Abteilung für Kardiologie Recruiting Bad Nauheim, Hessen, Germany, 61231 Contact: Helge Möllmann, MD +49 6032 996-2202 h.moellmann@kerckhoff-klinik.de Contact: Heike Wagner, Study Nurse +49 6032 996-5813 h.wagner@kerckhoff-forschungs-gmbh.de Principal Investigator: Helge Möllmann, MD Sub-Investigator: Christian Hamm, MD Universitätsmedizin Göttingen, Herzzentrum Recruiting Göttingen, Niedersachsen, Germany, 37099 Contact: Claudius Jacobshagen, MD +49 551-39-13239 jacobshagen@med.uni-goettingen.de Principal Investigator: Claudius Jacobshagen, MD Sub-Investigator: Wolfgang Schillinger, MD Herzzentrum Wuppertal Active, not recruiting Wuppertal, Nordrhein-Westfalen, Germany, 42117 Segeberger Kliniken GmbH Recruiting Bad Segeberg, Schleswig-Holstein, Germany, 23795 Contact: Gert Richardt, MD +49 4551 802 gert.richardt@segebergerkliniken.de Contact: Daniela Schürmann-Kuchenbrand, Study Nurse +49 4551 802 9947 daniela.schuermann-kuchenbrandt@segebergerkliniken.de Principal Investigator: Gert Richardt, MD Sub-Investigator: Ralph Ernst Tölg, MD Universitätsklinikum Schleswig-Holstein, Klinik für Innere Medizin III, Campus Kiel Not yet recruiting Kiel, Schleswig-Holstein, Germany, 24105 Contact: Norbert Frey, MD Contact: Petra Röthgen, SC +49 431 5971455 petra.roethgen@uk-sh.de Charité - Universitätsmedizin Berlin, Campus Benjamin Franklin Recruiting Berlin, Germany, 12203 Contact: Ulf Landmesser, MD ulf.landmesser@charite.de Contact: Anne-Christin Krämer, SN anne-christin.kraemer@charite.de Charité Universitätsmedizin Berlin, Campus Virchow-Klinik Recruiting Berlin, Germany, 13353 Contact: Florian Krackhardt, MD Florian.Krackhardt@charite.de Contact: Tina Gaulhofer, SC +49 30 450 553 782 christina.gaulhofer@charite.de Italy Careggi University Hospital, Invasive Cardiology Division Recruiting Firenze, Italy, 50141 Contact: David Antoniucci, MD david.antoniucci@virgilio.it Contact: Fabio Torrini, Assistant cardiologia1@aou-careggi.toscana.it Principal Investigator: David Antoniucci, MD Spaziani Hospital Frosinone Recruiting Frosinone, Italy, 03100 Contact: Maurizio Menichelli, MD +39 3382642317 menichelli747@yahoo.com Principal Investigator: Maurizio Menichelli, MD Collapse << |
| NCT02075268 | Healthy | Phase 1 | Unknown | February 2014 | Korea, Republic of ... more >> Asan Medical Center Recruiting Seoul, Korea, Republic of, 138-736 Contact: MI Jo Kim, MD 82-2-3010-4622 mjk0326@gmail.com Principal Investigator: Hyeong-Suk Lim, MD, PhD Collapse << |
| NCT01794000 | Sickle Cell Disease | Phase 3 | Terminated(The study is being ... more >>terminated for lack of efficacy.) Collapse << | - | - |
| NCT02215993 | Acute Coronary Syndrome ... more >> Platelet Function Collapse << | Phase 4 | Completed | - | Brazil ... more >> Federal University of São Paulo São Paulo, Brazil, 04024-002 Collapse << |
| NCT02545933 | Myocardial Infarction | Phase 4 | Recruiting | June 2019 | United States, Florida ... more >> University of Florida Recruiting Jacksonville, Florida, United States, 32209 Contact: Dominick J Angiolillo, MD, PhD dominick.angiolillo@jax.ufl.edu Principal Investigator: Dominick J Angiolillo, MD, PhD Collapse << |
| NCT01609647 | Acute Coronary Syndrome | Phase 3 | Completed | - | Korea, Republic of ... more >> DongA University Hospital Busan, Korea, Republic of, 602-715 Collapse << |
| NCT01587651 | - | - | Completed | - | - |
| NCT01543932 | Coronary Artery Disease | Phase 3 | Completed | - | Italy ... more >> Dept.of Cardiovascular Sciences,Policlinico Umberto I Rome, Italy, 000161 Collapse << |
| NCT02193971 | Acute Coronary Syndrome | Phase 4 | Recruiting | June 2020 | Korea, Republic of ... more >> Seoul National University Hospital Recruiting Seoul, Korea, Republic of, 110-744 Contact: Kyung Woo Park, MD, PhD 82-2-2072-0244 kwparkmd@snu.ac.kr Contact: Hyo-Soo Kim, MD, PhD 82-2-2072-2226 hyosoo@snu.ac.kr Collapse << |
| NCT01430091 | Sickle Cell Disease | Phase 1 | Completed | - | United States, Hawaii ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Honolulu, Hawaii, United States Collapse << |
| NCT01430091 | - | - | Completed | - | - |
| NCT01774838 | Acute Coronary Syndrome ... more >> Unstable Angina Collapse << | Phase 3 | Completed | - | Germany ... more >> Cardiology, University Hospital of Cologne Cologne, Germany, 50937 Collapse << |
| NCT02507323 | Cardiovascular Disease | Phase 2 | Withdrawn(collaborator withdre... more >>w the study) Collapse << | November 2017 | United States, Texas ... more >> Baylor-St. Lukes Medical Center Houston, Texas, United States, 77030 Houston Methodist Hospital Houston, Texas, United States, 77030 Memorial Hermann Hospital Houston, Texas, United States, 77030 Collapse << |
| NCT01790854 | Adverse Reaction to Antiplatel... more >>et Agent Acute Coronary Syndrome Collapse << | Phase 4 | Terminated(Low events rate. Sc... more >>arce economical resources) Collapse << | - | Italy ... more >> Careggi Hospital Florence, Italy Collapse << |
| NCT01591317 | Healthy Volunteers | Phase 1 | Completed | - | Korea, Republic of ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of Collapse << |
| NCT01365221 | Acute Coronary Syndrome | Phase 4 | Unknown | January 2014 | United States, Alabama ... more >> The Heart Center Huntsville, Alabama, United States, 35801 United States, District of Columbia Washington Hospital Center Washington, District of Columbia, United States, 20010 United States, Maryland Sinai Center for Thrombosis Research Baltimore, Maryland, United States, 21215 United States, Missouri Saint Luke's Hospital Kansas City, Missouri, United States, 64111 Collapse << |
| NCT01531114 | ST-Elevation Myocardial Infarc... more >>tion Collapse << | Phase 3 | Completed | - | Italy ... more >> Dept.of Cardiovascular Sciences,Policlinico Umberto I Rome, Italy, 00161 Collapse << |
| NCT01201772 | Coronary Artery Disease | Phase 4 | Completed | - | United States, Florida ... more >> University of Florida Jacksonville, Florida, United States, 32209 Collapse << |
| NCT01201772 | - | - | Completed | - | - |
| NCT01641510 | Acute Coronary Syndromes | Phase 3 | Recruiting | April 2018 | Korea, Republic of ... more >> DongA University Hospital Recruiting Busan, Korea, Republic of Contact: Moo Hyun Kim, MD +82-51-240-2976 kimmh@dau.ac.kr Principal Investigator: Moo Hyun Kim, MD Collapse << |
| NCT01979445 | Coronary Artery Disease (CAD) | Phase 2 | Completed | - | United States, Vermont ... more >> Fletcher Allen Health Care Burlington, Vermont, United States, 05401 Collapse << |
| NCT01979445 | - | - | Completed | - | - |
| NCT01014624 | Coronary Artery Disease | Phase 4 | Completed | - | United States, California ... more >> Scripps Clinic La Jolla, California, United States, 92037 United States, Florida University of Florida Health Science Center Shands Jacksonville Jacksonville, Florida, United States, 32209 United States, Ohio Medpace Clinical Pharmacology Unit Cincinnati, Ohio, United States, 45212 United States, South Dakota Black Hills Clinical Research Center Rapid City, South Dakota, United States, 57701 Collapse << |
| NCT01951001 | Acute Coronary Syndrome ... more >> Platelet Thrombus Bleeding Collapse << | Phase 4 | Unknown | October 2014 | Korea, Republic of ... more >> Gyeonsang National University Hospital Recruiting Jinju, Gyeonsangnam-do,, Korea, Republic of, 660-702 Contact: Young-Hoon Jeong, MD, PhD 82 10 7343 4670 goodoctor@naver.com Principal Investigator: Young-Hoon Jeong, MD, PhD Collapse << |
| NCT01869309 | Coronary Artery Disease | Phase 4 | Unknown | December 2016 | United States, Maryland ... more >> Sinai Center for Thrombosis Research Recruiting Baltimore, Maryland, United States, 21215 Contact: Kevin P Bliden, B.S. MBA 410-601-4795 kbliden@lifebridgehealth.org Contact: Tania B Gesheff, MSN 4106014795 tgesheff@lifebridgehealth.org Collapse << |
| NCT01684813 | Diabetes Mellitus Type II ... more >> Acute Coronary Syndrome Collapse << | Phase 4 | Completed | - | Spain ... more >> Hospital Universitario Virgen del Rocío Seville, Spain, 41013 Collapse << |
| NCT01876797 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT01014624 | - | - | Completed | - | - |
| NCT01789814 | - | - | Completed | - | - |
| NCT01115738 | Acute Coronary Syndrome | Phase 2 | Completed | - | India ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bangalore, India, 560099 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hyderabaad, India, 500 001 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. New Delhi, India, 110 060 Collapse << |
| NCT01591317 | - | - | Completed | - | - |
| NCT01115738 | - | - | Completed | - | - |
| NCT01178099 | Anemia, Sickle Cell | Phase 1 | Completed | - | United Kingdom ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. London, UK, United Kingdom, SE 1 1YR Collapse << |
| NCT01465828 | Acute Coronary Syndrome | Phase 3 | Completed | - | Italy ... more >> Dept.of Cardiovascular Sciences,Policlinico Umberto I Roma, Italy, 00161 Dept.of Cardiovascular Sciences,Policlinico Umberto I Rome, Italy, 00155 Collapse << |
| NCT01789814 | Coronary Artery Disease | Phase 4 | Completed | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT01178099 | - | - | Completed | - | - |
| NCT02075125 | - | - | Terminated(Enrolling participa... more >>nts has halted prematurely and will not resume) Collapse << | - | - |
| NCT02808767 | Myocardial Infarction ... more >> Angioplasty, Balloon, Coronary Platelet Aggregation Inhibitors Collapse << | Phase 4 | Completed | - | Czechia ... more >> Faculty Hospital Kralovske Vinohrady Prague, Czechia, 10034 Collapse << |
| NCT01648790 | - | - | Completed | - | - |
| NCT02075125 | ST-Segment Elevation Myocardia... more >>l Infarction Collapse << | Phase 3 | Terminated(Enrolling participa... more >>nts has halted prematurely and will not resume) Collapse << | - | Korea, Republic of ... more >> DongA University Hospital Busan, Korea, Republic of, 602-715 Collapse << |
| NCT01778842 | Coronary Artery Disease | Phase 3 | Unknown | July 2013 | Italy ... more >> Dept.of Cardiovascular Sciences,Policlinico Umberto I Not yet recruiting Rome, Italy, 000161 Contact: Rocco Stio, MD +300649979046 rocco.stio@libero.it Collapse << |
| NCT01835353 | Platelet Reactivity | Phase 3 | Completed | - | Greece ... more >> Dimitrios Alexopoulos Patras, Achaia, Greece, 26500 Collapse << |
| NCT02065479 | Coronary Artery Disease | Phase 4 | Recruiting | January 2020 | United States, Florida ... more >> University of Florida Recruiting Jacksonville, Florida, United States, 32209 Contact: Dominick Angiolillo dominick.angiolillo@jax.ufl.edu Principal Investigator: Dominick J Angiolillo, MD, PhD Collapse << |
| NCT01648790 | Healthy Volunteers | Phase 1 | Completed | - | United States, Texas ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dallas, Texas, United States Collapse << |
| NCT02016170 | Coronary Artery Disease ... more >> Acute Coronary Syndrome Collapse << | Not Applicable | Completed | - | United States, Florida ... more >> University of Florida Jacksonville, Florida, United States, 32209 Collapse << |
| NCT02016170 | - | - | Completed | - | - |
| NCT01560780 | Coronary Artery Bypass | Phase 3 | Completed | - | United States, California ... more >> San Francisco VA Medical Center, San Francisco, CA San Francisco, California, United States, 94121 United States, Florida North Florida/South Georgia Veterans Health System, Gainesville, FL Gainesville, Florida, United States, 32608 United States, Illinois Jesse Brown VA Medical Center Community-Based Outpatient Clinic Lake Side Divison, Chicago, IL Chicago, Illinois, United States, 60611 United States, Texas VA North Texas Health Care System Dallas VA Medical Center, Dallas, TX Dallas, Texas, United States, 75216 Collapse << |
| NCT01260584 | Coronary Artery Disease | Phase 4 | Completed | - | United States, Maryland ... more >> Sanai Center for Thrombosis Research Baltimore, Maryland, United States, 21215 United States, Ohio Medpace Clinical Pharmacology Unit Cincinnati, Ohio, United States, 45212 The Carl and Edyth Lindner Center for Research and Education at the Christ Hospital Cincinnati, Ohio, United States, 45219 Collapse << |
| NCT01852019 | Coronary Artery Disease | Phase 2 | Completed | - | United States, Vermont ... more >> Fletcher Allen Health Care Burlington, Vermont, United States, 05401 Collapse << |
| NCT01476696 | Sickle Cell Disease | Phase 2 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Oakland, California, United States, 94609 United States, District of Columbia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Washington, District of Columbia, United States, 20060 United States, Illinois For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chicago, Illinois, United States, 60614 United States, Louisiana For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. New Orleans, Louisiana, United States, 70112 United States, Massachusetts For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Boston, Massachusetts, United States, 02115 United States, Missouri For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. St Louis, Missouri, United States, 63104 United States, North Carolina For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chapel Hill, North Carolina, United States, 27599 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cincinnati, Ohio, United States, 45229 United States, Pennsylvania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT03387826 | Myocardial Infarction ... more >> Diabetes Mellitus Coronary Artery Disease Renal Disease Collapse << | Phase 4 | Recruiting | January 2019 | Greece ... more >> Attikon University Hospital Recruiting Chaidari, Greece, 12462 Contact: DIMITRIOS ALEXOPOULOS, MD 30-6944314398 dalex@med.uoa.gr Contact: Danai Sfantou, MD 6975023693 danaes8@yahoo.gr Collapse << |
| NCT01260584 | - | - | Completed | - | - |
| NCT01852019 | - | - | Completed | - | - |
| NCT01852175 | Coronary Artery Disease | Not Applicable | Completed | - | United States, Florida ... more >> University of Florida Jacksonville, Florida, United States, 32209 Collapse << |
| NCT01852175 | - | - | Completed | - | - |
| NCT01493999 | Acute Coronary Syndrome | Phase 4 | Completed | - | Hungary ... more >> University of Pécs, Heart Institute Pécs, Hungary, 7624 Collapse << |
| NCT03454841 | Acute Myocardial Infarction | Phase 4 | Recruiting | December 31, 2018 | Poland ... more >> Department of Cardiology, Wrocław Medical University Recruiting Wrocław, Dolnośląskie, Poland, 50-556 Contact: Wiktor Kuliczkowski, MD, PhD +48717842240 wkuliczkowski@wp.pl Principal Investigator: Wiktor Kuliczkowski, MD, PhD Sub-Investigator: Magdalena Cielecka-Prynda, MD Department of Cardiology, Dr. A. Jurasz University Hospital, Collegium Medicum, Nicolaus Copernicus University Recruiting Bydgoszcz, Kujawsko-pomorskie, Poland, 85-094 Contact: Piotr Adamski, MD, PhD +48525854023 piotr.adamski@wp.eu Principal Investigator: Aldona Kubica, PhD Sub-Investigator: Piotr Adamski, MD, PhD Sub-Investigator: Małgorzata Ostrowska, MD, PhD Collapse << |
| NCT03489863 | Coronary Artery Disease | Phase 4 | Recruiting | August 2019 | United States, Florida ... more >> University of Florida Recruiting Jacksonville, Florida, United States, 32209 Contact: Dominick J Angiolillo, MD, PhD dominick.angiolillo@jax.ufl.edu Principal Investigator: Dominick J Angiolillo, MD, PhD Collapse << |
| NCT02070159 | Coronary Artery Disease | Phase 3 | Completed | - | Korea, Republic of ... more >> DongA University Hospital Busan, Korea, Republic of, 602-715 Collapse << |
| NCT01852214 | Diabetes Mellitus ... more >> Coronary Artery Disease Collapse << | Not Applicable | Completed | - | United States, Florida ... more >> University of Florida Jacksonville, Florida, United States, 32209 Collapse << |
| NCT01852214 | - | - | Completed | - | - |
| NCT02212028 | - | - | Completed | - | - |
| NCT03672097 | Acute Coronary Syndrome (ACS) | Phase 4 | Recruiting | July 30, 2020 | Taiwan ... more >> Kaohsiung Veterans General Hospital Recruiting Kaohsiung, Taiwan Contact: See Central Contact China Medical University Hospital Recruiting Taichung, Taiwan Contact: See Central Contact Cheng Hsin General Hospital Recruiting Taipei, Taiwan Contact: See Central Contact Mackay Memorial Hospital Recruiting Taipei, Taiwan Contact: See Central Contact National Taiwan University Hospital Recruiting Taipei, Taiwan Contact: See Central Contact Tri-Service General Hospital Recruiting Taipei, Taiwan Contact: See Central Contact Collapse << |
| NCT02212028 | Coronary Artery Disease | Phase 4 | Completed | - | United States, Florida ... more >> University of Florida Jacksonville, Florida, United States, 32209 Collapse << |
| NCT01476696 | - | - | Completed | - | - |
| NCT03296540 | Cardiovascular Diseases | Phase 4 | Recruiting | April 1, 2021 | Netherlands ... more >> Erasmus Medical Center Recruiting Rotterdam, Netherlands, 3015 CE Contact: Roberto Diletti, MD, PhD r.diletti@erasmusmc.nl Maasstadziekenhuis Recruiting Rotterdam, Netherlands, 3079 DZ Contact: George Vlachojannis, MD, PhD +31685271550 VlachojannisG@maasstadziekenhuis.nl Collapse << |
| NCT02293395 | Acute Coronary Syndrome | Phase 2 | Completed | - | - |
| NCT00676520 | - | - | Completed | - | United States, California ... more >> Abbott Vascular Santa Clara, California, United States, 95054 Collapse << |
| NCT03243409 | - | - | Recruiting | August 31, 2017 | Spain ... more >> Hospital de la Santa Creu i Sant Pau Recruiting Barcelona, Spain, 08025 Contact: Laura Noguera, MD lnoguera@santpau.cat Collapse << |
| NCT02293395 | - | - | Completed | - | - |
| NCT02247128 | Aortic Valve Disease ... more >> Myocardial Infarction Stroke Bleeding Collapse << | Phase 4 | Recruiting | January 2020 | Belgium ... more >> Algemeen Stedelijk Ziekenhuis Recruiting Aalst, Belgium Contact: Ian Buysschaert, MD, PhD Ian.Buysschaert@asz.be Onze Lieve Vrouwe Ziekenhuis Recruiting Aalst, Belgium Contact: Bernard De Bruyne, MD, PhD bernard.de.bruyne@gammas.be Imelda Ziekenhuis Recruiting Bonheiden, Belgium Contact: John Roosen, MD John.Roosen@imelda.be Algemeen Ziekenhuis Sint Jan Recruiting Brugge, Belgium Contact: Luc Muyldermans, MD Luc.Muyldermans@azsintjan.be Ziekenhuis Oost-Limburg Recruiting Genk, Belgium Contact: Bert Ferdinande, MD Bert.Ferdinande@zol.be Universitair Ziekenhuis Leuven Recruiting Leuven, Belgium Contact: Christophe L.F. Dubois, MD, PhD christophe.dubois@uzleuven.be Czechia Charles university, Third Faculty of Medicine Recruiting Prague, Czechia Contact: Petr Tousek Luxembourg National Institute Surgery Cardiaque Et De Cardiologie Interventionnelle Recruiting Luxembourg, Luxembourg Contact: Peter Frambach, MD, PhD peter.frambach@incci.lu Netherlands Academic Medical Centre (AMC) Recruiting Amsterdam, Noord Holland, Netherlands, 1105 AZ Contact: Jan Baan, MD, PhD +31 20 566 9111 j.baan@amc.uva.nl Isala Clinics Recruiting Zwolle, Overijssel, Netherlands, 8011 JW Contact: Rik Hermanides, MD, PhD +31 38 424 5000 r.hermanides@isala.nl St. Antonius Hospital Recruiting Nieuwegein, Utrecht, Netherlands, 3435CM Contact: Jurrien M ten Berg, MD, PhD +31 88 320 1121 j.ten.berg@antoniusziekenhuis.nl Principal Investigator: Jurrien M ten Berg, MD, PhD Sub-Investigator: Vincent J Nijenhuis, MD Haga Ziekenhuis Recruiting Den Haag, Netherlands Contact: Carl Schotborgh, MD, PhD c.schotborgh@hagaziekenhuis.nl Medisch Spectrum Twente Recruiting Enschede, Netherlands Contact: Gert K van Houwelingen, MD, PhD K.vanHouwelingen@mst.nl University Medical Center Not yet recruiting Groningen, Netherlands, 9700 RB Contact: P van der Harst, MD, PhD +31 50 361 6161 Universitair Medisch Centrum Leiden Recruiting Leiden, Netherlands Contact: Frank van der Kley, MD F.van_der_Kley@lumc.nl Academic Hospital Recruiting Maastricht, Netherlands, 6229 HX Contact: Arnoud W.J. van 't Hof, MD, PhD +31 43 387 6543 arnoud.vant.hof@mumc.nl University Medical Center Utrecht (UMCU) Recruiting Utrecht, Netherlands, 3584 CX Contact: Pieter R Stella, MD, PhD +31 88 755 5555 p.stella@umcutrecht.nl Collapse << |
| NCT01134380 | - | - | Completed | - | France ... more >> Centre Hospitalier de la Région Annecienne Annecy, France Hôpital Albert Schweitzer Colmar, France, 68003 Centre Hospitalier Sud Francilien Corbeil Essonnes, France Centre Hospitalier d'Haguenau Haguenau, France Centre Hospitalier Saint Joseph et Saint Luc Lyon, France, 69007 Hôpital Privé Jacques Cartier Massy, France, 91300 Hôpital Bon Secours Metz, France Hôpital Arnaud de Villeneuve Montpellier, France Centre Hospitalier Universitaire Caremeau Nîmes, France Groupe Hospitalier Pitié Salpêtrière Paris, France Centre Hospitalier Rene Dubos Pontoise, France CHI de Villeneuve Villeneuve Saint Georges, France Collapse << |
| NCT02635230 | - | - | Active, not recruiting | December 2019 | Netherlands ... more >> Amphia Hospital Breda, Noord-Brabant, Netherlands, 4818 CK Collapse << |
| NCT00676520 | - | - | Completed | - | - |
| NCT03419325 | Cardiovascular Disease (CVD) ... more >> Stroke Acute Coronary Syndrome Peripheral Arterial Disease Coronary Artery Disease Myocardial Infarction Collapse << | Not Applicable | Recruiting | June 30, 2022 | Puerto Rico ... more >> University Hospital at Carolina Recruiting Carolina, Puerto Rico, 00984 Contact: Angel Lopez-Candales, MD candales33@gmail.com Cardiovascular Hospital of Puerto Rico and the Caribbean Recruiting San Juan, Puerto Rico, 00926 Contact: Dagmar F Hernandez-Suarez, MD dagmar.hernandez@upr.edu Collapse << |
| NCT01500434 | - | - | Completed | - | - |
| NCT00823212 | - | - | Completed | - | - |
| NCT01510327 | - | - | Completed | - | - |
| NCT01952392 | - | - | Completed | - | France ... more >> Research Site Paris, France Collapse << |
| NCT00823212 | Coronary Artery Disease | Phase 3 | Completed | - | - |
| NCT02886962 | Kidney Failure, Chronic | Phase 4 | Recruiting | January 2023 | France ... more >> Service de Néphrologie et médecine interne Not yet recruiting Amiens, France, 80000 Contact: Gabriel CHOUKROUN, MD CHRU d'Angers - Service de Néphrologie Not yet recruiting Angers, France, 49033 Contact: Virginie Besson, MD Service Néphrologie, Dialyse, Transplantation Not yet recruiting Caen, France, 14033 Contact: Patrick HENRI, MD Pathologie Rénale Recruiting Chambéry, France, 73000 Contact: Laure Croze, MD 0479965084 Service de Néphrologie Not yet recruiting Cherbourg-Octeville, France, 50102 Contact: Guillaume QUEFFEULOU, MD Aural Colmar Recruiting Colmar, France, 68000 Contact: Claire BORNI DUVAL, MD c.borni-duval@aural.asso.fr Service de néphrologie Not yet recruiting Colmar, France, 68000 Contact: Alexandre KLEIN, MD Service de néphrologie Not yet recruiting Dijon, France, 21000 Contact: Emmanuelle KOHLER, MD Aurad Aquitaine Not yet recruiting Gradignan, France, 33170 Contact: Catherine LASSEUR, MD Centre Hospitalier de Haguenau - service de Néphrologie Not yet recruiting Haguenau, France, 67504 Contact: Yves DIMITROV, MD AURAL Haguenau Not yet recruiting Haguenau, France Contact: Yves DIMITROV yves.dimitrov@ch-haguenau.fr Centre Hospitalier Emile Roux Not yet recruiting Le Puy-en-Velay, France, 43000 Contact: Marc BOUILLER marc.bouiller@ch-lepuy.fr Clinique Bouchard Recruiting Marseille, France, 13006 Contact: Jérome SAMPOL, MD Service de Néphrologie Not yet recruiting Nancy, France, 54511 Contact: Luc Frimat, MD ECHO de Nantes Recruiting Nantes, France, 44000 Contact: Angelo TESTA, MD Service de Néphrologie Not yet recruiting Nantes, France, 44093 Contact: Frédéric LAVAINNE, MD AURA Paris Plaisance Not yet recruiting Paris, France, 75014 Contact: Maxime TOUZOT, MD Hôpital Tenon - Service de Néphrologie Recruiting Paris, France, 75970 Contact: Hafedh FESSI, MD Service de néphrologie Recruiting Rennes, France, 35033 Contact: Cécile VIGNEAU, MD ECHO CA Laennec Not yet recruiting Saint-Herblain, France, 44821 Contact: Dan HRISTEA, MD NéphroCare Tassin-Charcot Not yet recruiting Sainte-Foy-lès-Lyon, France, 69110 Contact: Guillaume JEAN, MD AURAL st Anne Recruiting Strasbourg, France, 67000 Contact: Clotilde MULLER Clotilde-muller@hotmail.fr Service de Néphrologie Recruiting Strasbourg, France, 67000 Contact: Thierry Hannedouche, MD Centre Hospitalier Bretagne-Atlantique Not yet recruiting Vannes, France, 56017 Contact: Pierre Yves DURAND, MD Calydial CH Lucien Hussel Not yet recruiting Vienne, France, 38209 Contact: Agnès CAILLETTE BEAUDOIN, MD Collapse << |
| NCT01500434 | Coronary Artery Disease | Phase 3 | Completed | - | - |
| NCT01947998 | - | - | Recruiting | December 31, 2019 | United Kingdom ... more >> Recruiting Multiple Locations, United Kingdom Collapse << |
| NCT01103440 | Stable Angina | Phase 2 | Completed | - | United States, New York ... more >> Mount Sinai Medical Center New York, New York, United States, 10029 Collapse << |
| NCT01510327 | Coronary Artery Disease | Phase 3 | Completed | - | United States, Colorado ... more >> Medical Center of the Rockies (Loveland) Loveland, Colorado, United States, 80538 United States, Michigan Cardiac & Vascular Research Center of Northern Michigan Petoskey, Michigan, United States, 49770 Japan Saiseikai Yokohama-City Eastern Hospital Yokohama City, Kanagawa-ken, Japan Tokyo Women's Medical University Hospital Shinjuku-ku, Tokyo-to, Japan Sakakibara Heart Institute, Japan Research Promotion Society for Cardiovascular Diseases Fuchu-shi, Tokyo, Japan Collapse << |
| NCT01707927 | - | - | Recruiting | December 2022 | Norway ... more >> Oslo university hospital, Thoracic departement Recruiting Oslo, Norway Contact: Bjørn Braathen, MD. PhD 004722118080 uxjrbr@uus-hf.no Contact: Theis Tønnessen, MD, PhD 004722118080 uxthto@uus-hf.no Principal Investigator: Bjørn Braathen, MD-PhD Collapse << |
| NCT02402491 | Percutaneous Coronary Interven... more >>tion Collapse << | Phase 4 | Unknown | June 2015 | China, Beijing ... more >> Beijing Anzhen Hospital Beijing, Beijing, China, 100029 Collapse << |
| NCT01433978 | Idiopathic Thrombocytopenic Pu... more >>rpura Collapse << | Phase 3 | Terminated(Study was terminate... more >>d early due to significant enrollment challenges.) Collapse << | - | United States, South Dakota ... more >> Prairie Lakes Health Care System Watertown, South Dakota, United States Collapse << |
| NCT01947985 | - | - | Recruiting | December 31, 2019 | Netherlands ... more >> Recruiting Multiple Locations, Netherlands Collapse << |
| NCT01433978 | - | - | Terminated(Study was terminate... more >>d early due to significant enrollment challenges.) Collapse << | - | - |
| NCT01947959 | - | - | Active, not recruiting | December 31, 2019 | Germany ... more >> Multiple Locations, Germany Collapse << |
| NCT01438840 | - | - | Completed | - | - |
| NCT02118870 | Acute Coronary Syndrome | Phase 4 | Active, not recruiting | September 2018 | - |
| NCT01438840 | Chronic Thrombocytopenia ... more >> Immune Thrombocytopenia Collapse << | Phase 3 | Completed | - | Australia ... more >> Adelaide, Australia Bedford Park, Australia Belgium Antwerpen, Belgium Bulgaria Plovdiv, Bulgaria Sofia, Bulgaria Czechia Brno, Czechia Hradec Kralove, Czechia Praha, Czechia Netherlands Rotterdam, Netherlands New Zealand Christchurch, New Zealand Palmerston North, New Zealand Poland Bialystok, Poland Chorzow, Poland Gdansk, Poland Krakow, Poland Lodz, Poland Wroclaw, Poland Singapore Singapore, Singapore Slovakia Bratislava, Slovakia South Africa Johannesburg, South Africa Ukraine Dnipropetrovsk, Ukraine Donetsk, Ukraine Ivano-Frankivsk, Ukraine Kyiv, Ukraine Lviv, Ukraine Collapse << |
| NCT01385319 | Coronary Artery Disease | Phase 3 | Unknown | December 2017 | Hungary ... more >> Albert Szent-Györgyi Clinical Center, University of Szeged Szeged, Hungary Italy Ospedale San Donato Arezzo, AR, Italy, 52100 Ospedali Riuniti di Bergamo Bergamo, BG, Italy, 24128 Policlinico San Marco Zingonia, BG, Italy, 24040 Ospedale di Savigliano Savigliano, CN, Italy, 12038 Istituto Clinico Sant'Ambrogio Milano, MI, Italy, 20149 Azienda Unita' Sanitaria Locale Di Modena - Ospedale Baggiovara Modena, MO, Italy, 41100 Azienda Ospedaliero-Universitaria di Parma Parma, PR, Italy, 43126 Policlinico San Matteo Pavia, PV, Italy, 27100 Ospedale di Ravenna Ravenna, RA, Italy, 48121 Azienda Ospedaliera Ordine Mauriziano di Torino, Ospedale Umberto I Torino, TO, Italy, 10128 Ospedale San Giovanni Bosco Torino, TO, Italy, 10154 University Hospital of Ferrara Ferrara, Italy, 44100 Clinica Mediterranea Naples, Italy, 80122 Portugal Hospital de Santa Cruz Carnaxide, Portugal Switzerland University Hospital of Geneva Geneva, Switzerland Collapse << |
| NCT01103440 | - | - | Completed | - | - |
| NCT03606642 | Coronary Artery Disease ... more >> Atherosclerosis Stent Placement Collapse << | Phase 2 | Not yet recruiting | July 23, 2021 | United States, Arizona ... more >> HonorHealth Research Institute Not yet recruiting Scottsdale, Arizona, United States, 85258 Contact: Joanne Saczynski, RN 480-323-1046 Principal Investigator: David G Rizik, MD United States, Massachusetts Massachusetts General Hospital, Corrigan Minehan Heart Center Not yet recruiting Boston, Massachusetts, United States, 02114 Contact: Patricia Giunta 617-643-8769 pagiunta@mgh.harvard.edu Principal Investigator: Farouc Jaffer, MD, PhD Collapse << |
| NCT03062319 | Ischemic Stroke ... more >> Atrial Fibrillation Atherothrombosis Collapse << | Phase 4 | Recruiting | January 31, 2021 | Japan ... more >> Kobe City Medical Center General Hospital Recruiting Kobe, Hyogo, Japan, 650-0047 Contact: Nobuyuki Sakai, MD 078-302-7537 n.sakai@siren.ocn.ne.jp Contact: Nobuyuki Ohara, MD 078-302-7537 nobuyuki.ohara@gmail.com National Cerebral and Cardiovascular Center Recruiting Suita, Osaka, Japan, 565-8565 Contact: Hiroshi Yamagami, MD 06-6833-5012 ext 8025 yamagami-brain@umin.ac.jp Contact: Shuhei Okazaki, MD 06-6833-5012 ext 8123 s-okazaki@umin.ac.jp Collapse << |
| NCT03184805 | Coronary Artery Disease | Not Applicable | Terminated(Investigator judged... more >> that this study can not be maintained because participant registration rate is low.) Collapse << | - | Korea, Republic of ... more >> Division of Cardiology, Department of Internal Medicine, Yonsei University College of Medicine Seoul, Korea, Republic of, 03722 Collapse << |
| NCT01325116 | Acute Myocardial Infarction ... more >> STEMI Collapse << | Not Applicable | Completed | - | Canada, Ontario ... more >> Hamilton Health Sciences-General Site, Heart Investigation Unit Hamilton, Ontario, Canada, L8L 2X2 Collapse << |
| NCT02605447 | Coronary Artery Disease | Phase 4 | Active, not recruiting | August 2019 | - |
| NCT01626534 | Acute Coronary Syndrome | Phase 3 | Completed | - | France ... more >> Assistance Publique Hopitaux de Marseille Marseille, France, 13354 Collapse << |
| NCT01219894 | - | - | Withdrawn(Number of patients t... more >>o meet individual IDE requirements was not obtained.) Collapse << | - | - |
| NCT01121224 | Saphenous Vein Graft Atheroscl... more >>erosis Collapse << | Phase 4 | Completed | - | - |
| NCT01121224 | - | - | Completed | - | - |
| NCT00915733 | Myocardial Infarction | Phase 4 | Completed | - | Korea, Republic of ... more >> Gyeongsang National University Hospital Jinju, Korea, Republic of, 660-702 Collapse << |
| NCT01961856 | Platelet Reactivity | Phase 3 | Completed | - | Greece ... more >> Patras University Hospital Patras, Achaia, Greece, 26500 Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
